HAI, QUANG, BACH, BINH, CHOUDHARY, and LEE: Tripterygium wilfordii (Celastraceae): A new generic and species record for the flora of Vietnam
Abstract
Tripterygium wilfordii Hook.f. (Celastraceae), collected from Phia Oac-Phia Den National Park of Vietnam, is reported here as a new generic and species record for the flora of Vietnam. The genus Tripterygium differs from other genera of Celastraceae by its samara fruits, which become three-winged at maturity, and a scandent shrub. Taxonomic notes, descriptions, line drawing and photographs are provided, together with short notes on the distribution, ecology and phenology of Tripterygium wilfordii.
Keywords: protected area, Phia Oac-Phia Den, floristic diversity, range extension
The Celastraceae ( Brown, 1814: 554) family of angiosperms consists of almost 100 genera and approximately 1,350 species ( Simmons, 2004) in the world. Southeast Asia is one of the main centres of diversity of the family. A recent checklist of Celastraceae in Vietnam documented 13 genera and about 80 species under the family ( Ban, 2003). Thereafter, several new genera and species were documented for the family in Vietnam ( Simmons, 2004; Chinh et al., 2016; Savinov et al., 2018, 2019), taking the total count to 16 genera and about 83 species in the country. Phia Oac-Phia Den National Park of the Cao Bang province in northern Vietnam shares its border with China, and known for its rich biodiversity ( Fig. 1). Being located at the intersection of the tropical and subtropical climatic zones, the National Park provides a suitable habitat for many interesting flora and fauna. In 2016, we made a floristic survey of this area, and collected an interesting plant species. After a thorough scrutiny of the taxonomic literature and type specimens housed in several herbaria (HN, HNU, K, P, VNM), the collected species was identified as Tripterygium wilfordii Hook.f. which was never reported from Vietnam. Tripterygium wilfordii was described by Hooker (1862), based on the material collected by Wilford in 1858 from Taiwan (Formosa). Presently, the genus Tripterygium is known by only four species in the world, i.e. T. doianum, T. hypoglaucum, T. regelii, and T. wilfordii ( POWO, 2021). However, their traditional morphological circumscription remains in question. Some recent studies vouched for their delimitation to a single species exhibiting extreme variation in their morphology ( Ma et al., 1999; Simmons, 2004). There have been conflicting view points of the taxonomists based on their treatments based on morphological and molecular data. Ma and Funston (2008) pointed out the difficulty to distinguish Tripterygium hypoglaucum and T. regelii based on morphological data and they synonymized them under T. wilfordii. The molecular studies conducted by Law et al. (2011) did not consider T. hypoglaucum and T. doianum distinct from T. wilfordii. The study also recognised T. regelii as a distinct species. In the molecular study conducted by Ma et al. (2017), T. wilfordii and T. hypoglaucum clustered together and did not appear as distinct lineages, while T. regelii nested out as a separate cluster. Authors of the present work also believe that T. hypoglaucum is conspecific with T. wilfordii, while T. regelii deserves a distinct species status. The genus Tripterygium clearly differs from other Celastraceae genera in Vietnam by having three lateral winged samaras in contrast to the other genera having drupe, berry, and capsule types of fruits. The genus was hitherto known to occur in China, Japan, Korea, and Myanmar ( Simmons, 2004). The present report is a new distributional record of this genus in Vietnam. We provide a description, line drawing and colour photographs of Tripterygium wilfordii, with an updated key to the genera of Celastraceae in Vietnam for easy identification.
Materials and Methods
The newly recorded species was collected during an expedition by the scientists from Vietnam and Korea in the Phia Oac-Phia Den National Park in Vietnam during June 2016. The specimens collected from the natural habitats were thoroughly processed using standard herbarium techniques ( Jain and Rao, 1977) and deposited in the Herbarium of Institute of Ecology & Biological Resources (HN) Vietnam and Chungnam National University, Korea. Morphological characters were studied consulting the relevant literature ( Hou, 1962; Lu and Yang, 1993; Cheng, 1999; Ma et al., 1999; Ho, 2000; Simmons, 2004; Ma and Funston, 2008). The specimens of Tripterygium and allied genera kept in different herbaria of Vietnam such as Herbarium of Institute of Ecology and Biological Resources (HN), VNU University of Science (HNU), Institute of Tropical Biology (VNM) were studied. Photographs of the specimens housed in the virtual herbaria such as K, P were also studied.
Taxonomic Treatment
Tripterygium Hook.f. in Benth. & Hook.f., Gen. Pl. 1: 368, 1862.—TYPE: Tripterygium wilfordii Hook.f.
Scandent shrubs or sometimes semi-woody vines, glabrous or tomentose; branchlets sometimes 4–6-angled. Leaves alternate, serrate, stipulate. Inflorescence axillary or terminal, thyrsoid. Flowers bisexual or unisexual (polygamous), 5-merous. Disk fleshy, cupuliform, 5-lobed, intrastaminal; anthers longitudinally dehiscent, introrse. Ovary 3-locular; ovules erect, 2 per locule. Samara 3-winged, chartaceous. Seed 1, trigonous, albuminous.
Distribution: China, Japan, Korea, Taiwan ( Simmons, 2004), and Vietnam.
Tripterygium wilfordii Hook.f. in Benth. & Hook.f., Gen. Pl. 1: 368, 1862 ( Figs. 2, 3).—TYPE: Taiwan. NW Formosa, June 1858, C. Wilford 484 (Lectotype or possibly holotype, K, [2 sheets], K000478771, K000478770, photos! [ Turner, 2016]).
Aspidopterys hypoglauca H. Lév., Repert. Spec. Nov. Regni Veg. 9: 458, 1911; Tripterygium hypoglaucum (H. Lév.) Hutchins., Bull. Misc. Inform. Kew: 101, 1917.—TYPE: China. Kouy-Tcheou (Guizhou), Ma-Jo, J. Cavalerie 3316 (photo, https://plants.jstor.org/search).
Deciduous subshrubs, or scandent and scrambling, or sometimes semi-woody vines, 2–4 m; branching distally, slender or scrambling, minutely pubescent when young or in open sunlight, becoming glabrous when old or in shade, slightly angled, verrucose or not. Stipules linear, caducous. Leaves simple; petiole 1–1.5 cm; blade usually ovate or rounded-ovate, sometimes oblong or elliptic-ovate, 5–8 × 3–6 cm, papery, leathery, glabrous or sparsely scurfy tomentose with reddish brown hairs, abaxially ± farinose, base broadly cuneate, or rounded to cordate, margin entire or less often crenulate, apex short to long acuminate or acute, tip often blunt. Thyrses large, composed of several to dozens of thyrslets, 10–15 × 4–8 cm; flowers 1 normal and 1 unfertilized male. Flowers whitish, greenish, or yellow-green, small, 4–6 × 4–6 mm; calyx 5-lobed, hemispheric, ca. 1 mm; petals 5, oblong to subovate, slightly narrowed to base, 2–2.5 mm, apex rounded; stamens 5, inserted at margin of cup-shaped and compressed disk; disk bright green, fleshy, ca. 2 mm in diam; ovary superior, 3-edged, combined with disk at base, incompletely 3-locular, with 3 prominent ca. 1 mm lobes, short style at apex; stigma capitate, bright purple. Samara usually green or greenish brown when mature, sometimes pink or pinkish purple, 1.5–2.0 × 1.0–1.5 cm. Seed compressed, 3-angled, ca. 5 × 1.5–3 mm.
Flowering and fruiting: May to October.
Distribution: Vietnam: Cao Bang Province, Nguyen Binh District, Quang Thanh commune. China, Japan, Korea, Myanmar and Taiwan.
Ecology and habitat: Tripterygium wilfordii was found growing along the roadside forest margins at about 1,122 m above sea level.
Specimens examined: VIETNAM. Cao Bang Province: Nguyen Binh district, Quang Thanh commune, Phia Oac-Phia Den National Park, 29 June 2016, near 22° 37′ 32.0″ N, 105° 52′ 52.4″E; 1,122 m a.s.l., T. T. Bach, V. T. Chinh, D. V. Hai, D. H. Son, T. D. Binh, Joongku Lee, Sang-Jin Lee, Donghyeong Kwak, Hyun Jae Cho, HNCNU 318 (HN, herbarium of Chungnam National University).
Tripterygium wilfordii is currently known from one locality in Nguyen Binh District of Cao Bang Province of North Vietnam: Quang Thanh Commune. The plant grows along the road, forest margins of secondary evergreen broad-leaved forest in association with Antidesma bunius, Boenninghausenia albiflora, Mallotus barbatus, Saurauia rubricalyx, and Trema tomentosa at an elevation of 1,100–1,200 m a.s.l. During our investigation, we could find only one population growing under secondary forests. Therefore, the information currently obtained is inadequate to assess the species risk of extinction, whether direct or indirect. Further field investigation is needed, and it is expected that the species may be found in other localities from Vietnam. In accordance to the IUCN Red List Categories (2012, ver 3.1), we propose a temporary listing of this species as a taxon under the Data Deficient (DD) category.
Key to the genera of Celastraceae in Vietnam
(partly based on Hou, 1962; Hou et al., 2010; Simmons, 2004)
Stamens numerous ············································································································································· 1. Plagiopteron Stamens 2–5.
2. Disk intrastaminal or stamens on disk; flowers unisexual or bisexual; fruit drupaceous, baccate, samaroid, or capsular; seeds with or without surrounding wings, arillate or exarillate.
4. Leaves decussate or opposite.
9. Petals slightly united at the base. Disk proper absent, filaments united at the base in a ring or short tube, by some interpreted to represent a disk, usually united with the petals ·········································· 7. Microtropis 9. Petals free. Disk conspicuous, fleshy, cupular or flat, free from the petals.
10. Ovary 3-, or 4–5 celled. Ovules 1–18 in each cell.
10. Ovary 2-celled, or 1-celled by abortion. Ovules 2 in each cell.
13. Disk more or less flat. Anthers subglobose and rounded at the apex, connective invisible on the dorsal side ·········································································································································· 11. Cassine 13. Disk cupular. Anthers ovoid and short-apiculate, connective distinct and broad on the dorsal side ············································································································································· 12. Pleurostylia
3. Plants with thorns or stems terminating in sharp points ·································································· 13. Gymnosporia
2. Disk extrastaminal; flowers bisexual; fruit baccate with seeds surrounded by mucilaginous arils, transversely-flattened, trilobed capsules with seeds with membranous basal wings or narrow stipes, or capsules that are circular or trigonous in cross section with seeds with membranous apical wings or arils basal to partially enveloping seeds.
14. Fruit baccate with seeds surrounded by mucilaginous arils ······································································ 14. Salacia 14. Fruit transversely-flattened, trilobed capsules with seeds with membranous basal wings or narrow stipes, or capsules that are circular or trigonous in cross section.
15. Ovules 2 per locule ·························································································································· 15. Reissantia 15. Ovules 3–22 per locule ················································································································ 16. Loeseneriella
ACKNOWLEDGMENTS
Authors are grateful to the curators of herbaria HN, VNM, and HNU. This research was supported by the Bio & Medical Technology Development Program of the National Research Foundation (NRF), South Korea funded by the Korean government (MSIT) (NRF-2017M3A9A507020221). The fourth author (RKC) thanks the Director, Agharkar Research Institute for facilities and encouragements.
Fig. 1.
Distribution map of Tripterygium wilfordii in northern Vietnam. 
Fig. 2.
Tripterygium wilfordii Hook.f. A. Branchlet with inflorescence. B. A part of the inflorescence. C. Branchlet with infructescence. D. Fruits. 
Fig. 3.
Tripterygium wilfordii Hook.f. A. Branchlet with inflorescence. B. A part of leaf. C. A part of inflorescence. D. Flower. E. Stamen. F. Ovary and style. G. Fruit (Line drawing by Ms. Le Kim Chi). 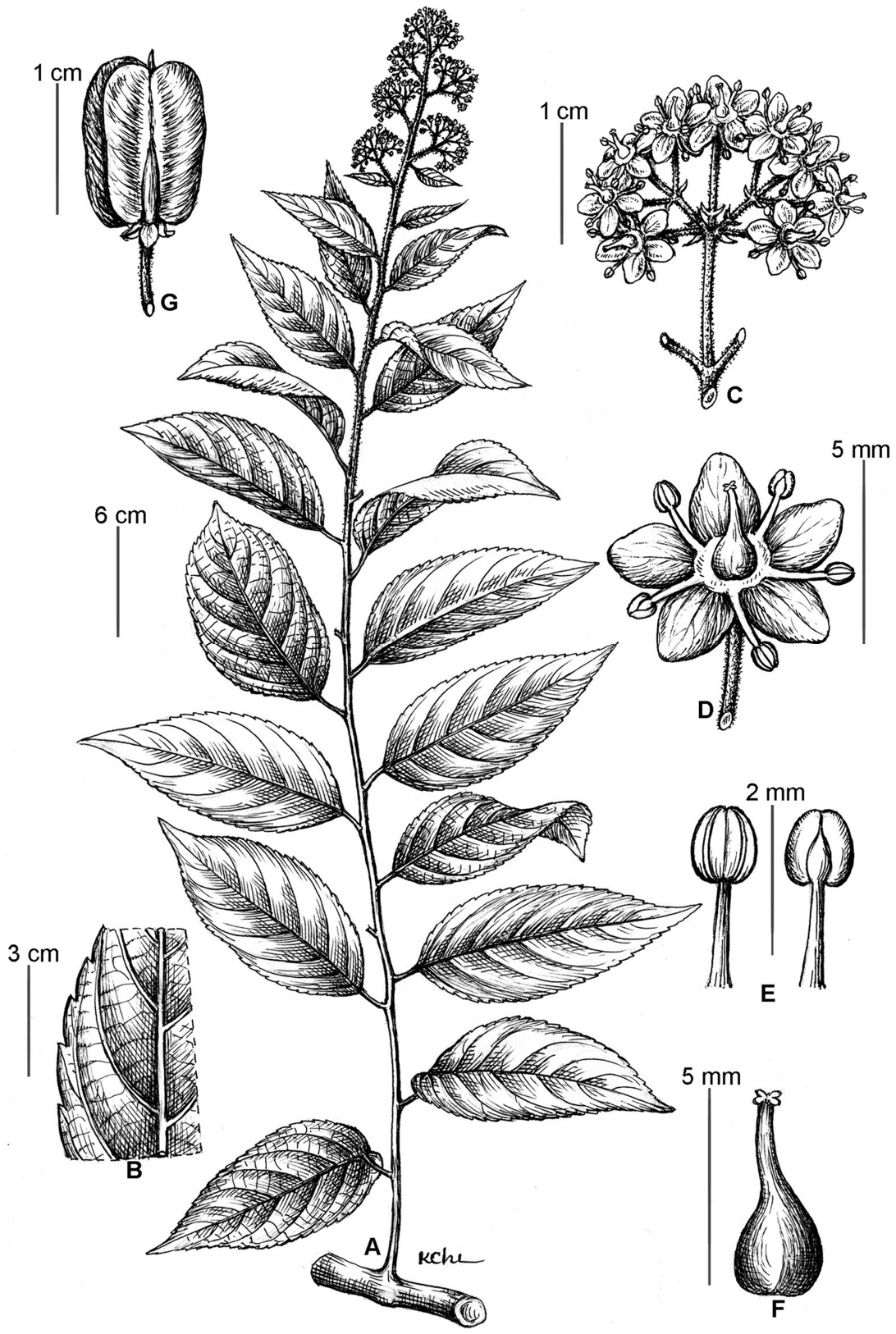
Literature Cited
Ban, NT. 2003. Celastraceae. In Checklist of Plant Species of Vietnam. 2: Ban, NT (ed.), Agricultural Publishing House, Hanoi. 1120-1134 (in Vietnamese).
Brown, R. 1814. Celastraceae. In A Voyage to Terra Australis 2. Flinders, M (ed.), W Bulmer and Co, Cleveland-Row, London. 554 pp.
Cheng, CY. 1999.
Tripterygium. In Flora Republicae Popularis Sinicae 45(3). Cheng, CY. Huang, PH (eds.), Science Press, Beijing. 178-181 (in Chinese).
Chinh, VT. Choudhary, RK. Bach, TT. Quang, BH. Hai, DV. Eum, S and Lee, J. 2016. Morphological and anatomical studies of the newly recorded genus Plagiopteron Griff. (Celastraceae) from Vietnam. Bangladesh Journal of Plant Taxonomy 23: 161-166.  Ho, PH. 2000. Celastraceae. In Cây Cỏ Việt Nam 2. Tre Publishing House, Ho Chi Minh. 142-162 (in Vietnamese).
Hooker, JD. 1862.
Tripterygium. In Genera Plantarum ad Exempla Imprimis in Herbariis Kewensibus Servata Definita. I: Pars I. Bentham, G. Hooker, JD (eds.), London. 368 pp.
Hou, D. 1962. Celastraceae. In Flora Malesiana, Ser. I, 6(2). Van Steenis, CGGJ (ed.), Wolters-Noordhoff Publishing, Groningen. 227-291.
Hou, D. Savinov, IA and Van Welzen, PC. 2010. Celastraceae. In Flora of Thailand. 10(2): Santisuk, T. Larsen, K (eds.), Prachachon Co Ltd, Bangkok. 141-198.
IUCN. 2012. IUCN Red List Categories and Criteria. Version 3.1. 2nd ed. IUCN, Gland and Cambridge. 32 pp.
Jain, SK and Rao, RR. 1977. A Handbook of Field and Herbarium Methods. Today and Tomorrow. Printers and Publishers, New Delhi. 157 pp.
Law, SK-Y. Simmons, MP. Techen, N. Khan, IA. He, M-F. Shaw, P.-C and But, PP-H. 2011. Molecular analyses of the Chinese herb Leigongteng ( Tripterygium wilfordii Hook. f.). Phytochemistry 72: 21-26.   Lu, SY and Yang, YP. 1993. Celastraceae. In Flora of Taiwan. 2nd ed. 3: National Science Council of the Republic of China, Taipei. 640-660.
Ma, B. Hu, T. Li, P. Yuan, Q. Lin, Z. Tu, Y. Li, J. Zhang, X. Wu, X. Wang, X. Huang, L and Gao, W. 2017. Phylogeographic and phylogenetic analysis for Tripterygium species delimitation. Ecology and Evolution 7: 8612-8623.    Ma, J.-S. Brach, AR and Liu, Q.-R. 1999. A revision of the genus Tripterygium (Celastraceae). Edinburgh Journal of Botany 56: 33-46.
Ma, JS and Funston, AM. 2008.
Tripterygium. In Flora of China. 11: Oxalidaceae through Aceraceae. Wu, ZY. Raven, PH. Hong, DY (eds.), Science Press, Beijing and Missouri Botanical Garden, St Louis, MO. 486-487.
Savinov, IA. Nuraliev, MS. Vislobokov, NA. Pan, B. Wen, F. Fu, L.-F. Wei, Y.-G and Averyanov, LV. 2018. Lectotypification and new data on distribution of Glyptopetalum sclerocarpum (Celastraceae). Phytotaxa 350: 33-41.  Savinov, IA. Nuraliev, MS. Kuznetsov, AN. Kuznetsova, SP. Luu, HT. Tran, HD and Luong, HT. 2019.
Microtropis cerocarpa (Celastraceae), a new species from southern Vietnam. Phytotaxa 387: 140-148.  Simmons, M. 2004. Celastraceae. In The Families and Genera of Vascular Plants. 6: Kubitzki, K (ed.), Springer-Verlag, Berlin. 29-64.
Turner, IM. 2016. Species names attributed to Bentham & Hooker’s Genera Plantarum. Annales Botanici Fennici 53: 285-337. 
|
|














